TPL Path Labs offers services both for your preferred tissue biomarker and the characterisation of novel large molecules such as therapeutic monoclonal antibodies. For this purpose, we are capable of providing the full service required for executing Tissue Cross Reactivity (TCR) studies.
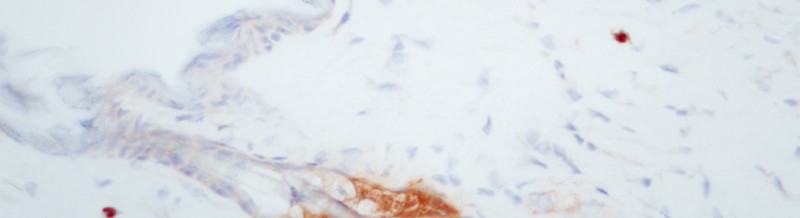
CD34 Tissue Cross Reactivity validation study (mouse skin)
To offer customised TCR services with validated study-specific protocols reflecting the regulator´s exact requirements and your needs in a cost- and time-efficient manner, we use a three phase approach.
PHASE 1: Initial assay and protocol optimisation
PHASE 2: Optional preliminary non-GLP TCR Screening
PHASE 3: Full GLP cross reactivity study
After screening a limited tissue panel in phase 2, the main GLP-compliant TCR study is performed on a panel of 38 human tissues/organs from a minimum of three donors as required by both, FDA and EMA.
